Executive Summary
Above cuff vocalization (ACV) enables selected patients with a cuffed tracheostomy tube to produce voice while the cuff remains inflated through delivery of a controlled flow of medical air or oxygen via the speech lumen. Loss of verbal communication following tracheostomy is associated with significant patient distress, reduced quality of life, and impaired participation in care. Many patients are unable to tolerate cuff deflation or one-way speaking valves due to ventilatory dependence or airway instability, limiting access to traditional voicing strategies.
This evidence-informed practice guidance was developed to provide standardized, actionable guidance for the safe implementation of ACV across acute and post-acute care settings. The guidance synthesizes peer-reviewed evidence and interprofessional clinical expertise, and is reported in alignment with the RIGHT (Reporting Items for Practice Guidelines in Healthcare) statement. Recommendations emphasize structured patient selection, clearly defined interprofessional roles, low-flow gas flow titration, time-limited use, and explicit safety safeguards to mitigate known risks such as air trapping, mucosal injury, and equipment misconnection.
The guidance is intended for use by speech-language pathologists, respiratory therapists, nurses, physicians, and advanced practice providers caring for adults with tracheostomy in intensive care units, step-down units, and long-term acute care settings. Adoption of standardized ACV practices has the potential to facilitate earlier return of speech, improve patient experience, and enhance communication while maintaining airway safety.
Guidance Development and Reporting Framework
This practice guidance is evidence-informed and implementation-focused. It synthesizes peer-reviewed literature and interprofessional clinical expertise to support safe clinical use of ACV. It is not intended to replace formal society-endorsed consensus guidelines where available. The manuscript was developed with reference to the RIGHT statement to enhance transparency in reporting.1 Evidence was identified through narrative review of the literature addressing ACV mechanisms, safety, communication outcomes, and implementation in acute and post-acute care settings. Evidence was mapped to specific practice recommendations to support transparency and reproducibility.
Evidence informing this guidance was identified through a structured narrative review of the literature conducted between January 2000 and January 2026 using PubMed, CINAHL, Scopus, and Google Scholar. Search terms included combinations of above cuff vocalization, talking tracheostomy tube, communication in mechanically ventilated patients, tracheostomy communication, and subglottic airflow speech. Peer-reviewed studies describing ACV mechanisms, safety outcomes, implementation protocols, and communication outcomes in adults with tracheostomy were prioritized. Observational studies, implementation reports, systematic and narrative reviews, and relevant physiologic investigations were included to capture the breadth of available evidence. Evidence was prioritized based on clinical relevance to ACV safety, feasibility, and communication outcomes, and was mapped to specific recommendations to enhance transparency.
Formal evidence grading systems (e.g., Grading of Recommendations Assessment, Development and Evaluation [GRADE]) were not applied, as the current evidence base consists primarily of observational studies, implementation reports, and narrative reviews. Recommendations are therefore evidence-informed and consensus-supported rather than strength graded; intended to support clinical decision-making while allowing adaptation to local context and resources.
Background and Rationale
Tracheostomy is a commonly performed intervention in critically ill patients who require prolonged mechanical ventilation or airway protection.2 Although tracheostomy can facilitate ventilator weaning, airway clearance, and long-term respiratory support, inflation of the tracheostomy cuff prevents airflow through the upper airway and vocal folds, resulting in aphonia and significant communication impairment.3
Loss of verbal communication following tracheostomy has been consistently identified as a major source of patient distress and reduced quality of life, contributing to anxiety, frustration, delirium, and impaired participation in care.4,5 A systematic review further describes anxiety, frustration, loss of control, and difficulty expressing basic needs as common emotional experiences among patients who cannot communicate verbally during critical illness.6 Restoration of voice has been associated with improved patient experience, enhanced engagement with clinicians and family members, and improved perceived quality of life in patients with tracheostomy.7 However, many patients remain unable to tolerate cuff deflation or one-way speaking valves due to ventilatory dependence, aspiration risk, or airway instability, limiting access to traditional voicing strategies.8
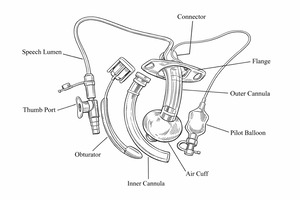
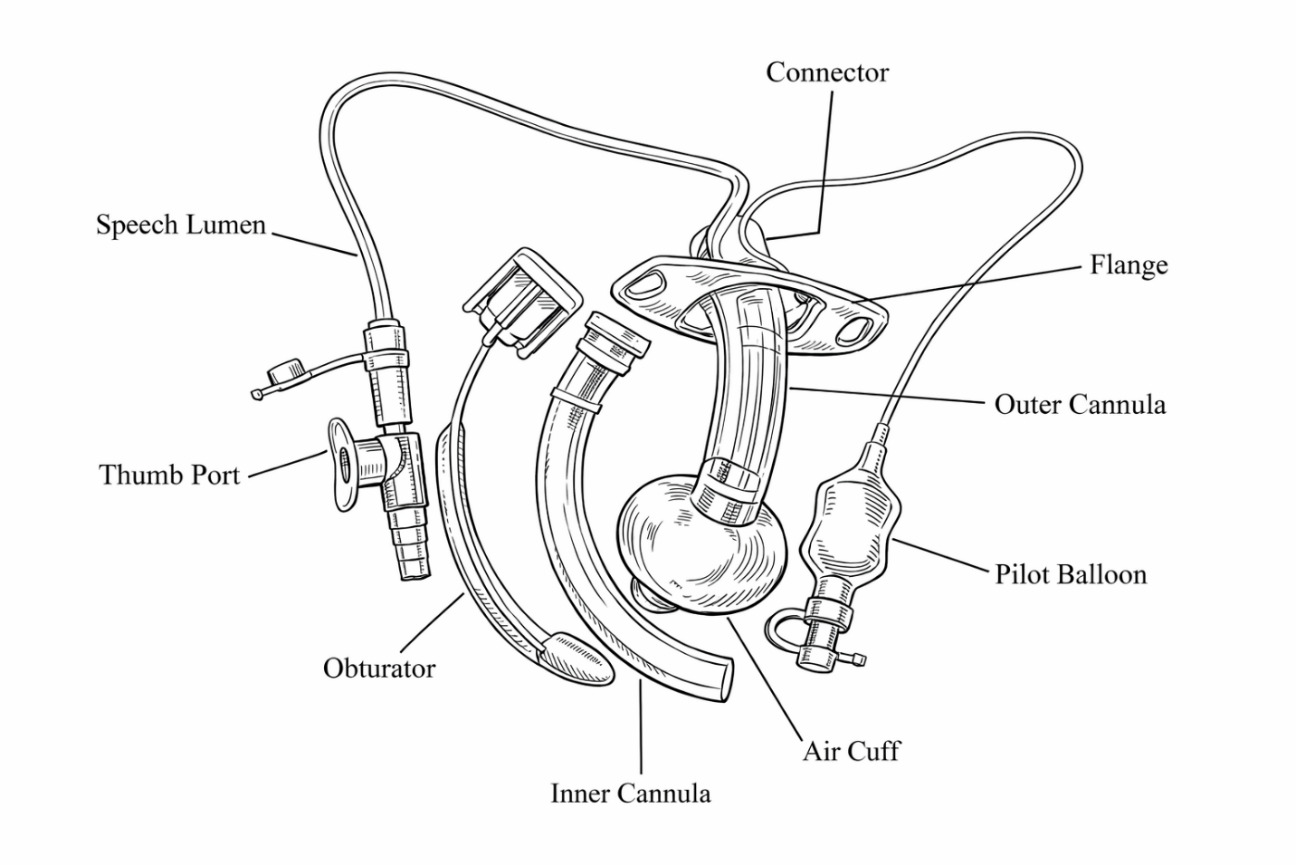
ACV is a communication technique that enables phonation while the tracheostomy cuff remains inflated by delivering a controlled flow of medical air or oxygen through the subglottic suction port (speech lumen) of an ACV-capable tracheostomy tube (Figure 1).4,8 ACV is the only voicing method that permits speech with the cuff fully inflated, making it uniquely applicable to patients who require ongoing mechanical ventilation or airway protection.4 Above cuff vocalization was first described in 1967 by Whitlock and further discussed in 1975 by Safar and Grenvik as a means of enabling speech in patients with cuffed tracheostomy tubes. More recently, the technique has been refined through contemporary protocols and interprofessional implementation efforts, particularly in the United Kingdom and Australia, with growing attention to its feasibility, safety, and clinical utility in modern critical care settings.8–11
An expanding body of evidence demonstrates that ACV can facilitate earlier return of speech, with successful phonation achieved in approximately 80–90% of appropriately selected patients in published cohorts9,12 and low rates of serious adverse events when implemented within structured protocols.12,13 Hospital-wide implementation studies have shown that standardized ACV protocols are feasible across diverse intensive care unit populations and are associated with reduced time to first speech following tracheostomy placement.12,13 Additional benefits reported in the literature include improvements in patient-reported ease of communication and overall communication effectiveness during critical illness.7
Despite growing evidence supporting ACV, its adoption remains inconsistent across institutions and healthcare systems. International surveys indicate that fewer than half of clinical services report routine ACV use, with variability in clinician familiarity, training, and protocol availability.14 Reported risks include subglottic air trapping, mucosal drying, laryngospasm, stomal air leak, and equipment-related errors, including inadvertent misconnection of airflow to the pilot balloon inflation port.4,15 Although major complications are uncommon, these risks underscore the importance of protocolized implementation, competency-based training, and explicit safety safeguards.16
Evidence further supports the importance of an interprofessional approach to communication interventions in patients with tracheostomy, with speech-language pathologists, respiratory therapists, nurses, and physicians contributing complementary expertise in airway assessment, ventilation management, communication coaching, and patient monitoring.5,8 Multidisciplinary, speech-language pathology-led protocols have been shown to improve feasibility, consistency, and clinician confidence while minimizing preventable adverse events.12
Given the expanding evidence base, persistent variability in clinical practice, and the high stakes associated with airway safety and patient communication, there is a clear need for standardized, evidence-informed practice guidance that translates existing literature into actionable clinical practice.2,7 This guidance was developed to address this gap by providing standardized criteria for patient selection, delineation of interprofessional roles, structured procedures, and explicit safety and monitoring strategies to support safe and consistent implementation of ACV across acute and post-acute care settings.
Aim and Objectives
The aim of this guidance is to provide standardized, evidence-informed guidance for the safe initiation, implementation, monitoring, and discontinuation of ACV in patients with tracheostomy.
Specific objectives are to:
-
Support earlier and safer restoration of verbal communication in selected patients with tracheostomy.
-
Reduce practice variability through standardized criteria and procedures.
-
Minimize preventable adverse events through explicit safety safeguards.
-
Clarify interprofessional roles and accountability.
Target Population, End Users, and Care Settings
Target population: Adult patients with a cuffed tracheostomy tube who require the cuff to remain inflated for ventilation or airway protection and who are awake, alert, and attempting to communicate. Pediatric populations were not included in this guidance due to limited available evidence regarding the safety and implementation of ACV in children.
End users: Speech-language pathologists, respiratory therapists, nurses, physicians, and advanced practice providers.
Care settings: Intensive care units, step-down units, and long-term acute care hospitals.
Guidance Statement
ACV is a communication intervention that enables selected patients with a cuffed tracheostomy tube to produce voice while the cuff remains inflated. This practice guidance supports the safe, evidence-informed use of ACV through a standardized, interprofessional approach to optimize patient communication, quality of life, and participation in care. This guidance outlines clear accountability, competency expectations, and risk mitigation strategies consistent with Magnet standards, Joint Commission patient safety goals, and institutional high-reliability principles.
Purpose
The purpose of this guidance is to establish standardized criteria, roles, procedures, and safety measures for the initiation, implementation, monitoring, and discontinuation of ACV in hospitalized patients with a tracheostomy.
Scope
This guidance applies to all inpatient care areas where patients with tracheostomy tubes are managed, including intensive care units, step-down units, and long-term acute care settings. It is intended for use by the interprofessional team, including physicians, advanced practice providers, nurses, respiratory therapists, and speech-language pathologists.
Although this guidance is written in a format compatible with institutional policy development, the recommendations are intended to be broadly applicable to clinical practice across healthcare systems. Institutions may adapt these recommendations to local workflows, staffing models, equipment availability, and regulatory requirements while maintaining the core safety principles outlined in this document.
Definitions
-
Above Cuff Vocalization: Delivery of a controlled flow of gas (medical air or oxygen) through the speech lumen of a tracheostomy tube with an inflated cuff to facilitate gas flow through the vocal folds and enable phonation.
-
ACV-capable Tracheostomy Tube: A cuffed tracheostomy tube with a speech lumen designed to allow suctioning of subglottic secretions and delivery of gas flow for ACV.
Indications
ACV may be considered for patients who meet all of the following criteria:
-
Presence of a cuffed tracheostomy tube with a speech lumen
-
Inflated cuff required for mechanical ventilation or airway protection
-
Awake, alert, and attempting to communicate
-
Hemodynamically stable
-
Adequate upper airway patency as determined by the clinical team
-
Ability to tolerate brief periods of ACV during supervised trials
-
Ability to regulate gas flow delivery for phonation, either independently or with assistance from a trained individual (e.g., clinician, family member, or caregiver)
Contraindications
ACV should not be initiated in patients with any of the following:
-
Fresh tracheostomy < 72 hours since tracheostomy placement
-
Known or suspected upper airway obstruction or laryngectomy
-
Unstable tracheostomy position or concern for malposition
-
Active subcutaneous emphysema or high risk for air leak into tissues
-
Severe autonomic instability or uncontrolled arrhythmias
-
Nonverbal baseline status due to pre-existing neurologic or structural conditions
-
Copious bleeding from the tracheostomy or stoma site
Interprofessional Roles and Responsibilities
Speech-Language Pathology (SLP)
-
Lead patient assessment for communication readiness
-
Determine candidacy for ACV
-
Conduct initial ACV trial and assess voice quality, intelligibility, and patient tolerance
-
Provide patient training and education
-
Develop recommendations for ongoing ACV use
-
Determine minimal gas flow for optimal voicing
-
Label suction line to reduce risk of misconnection to cuff
Respiratory Therapy
-
Verify tracheostomy tube type and integrity
-
Assist with setup of gas flow delivery for ACV
-
Monitor respiratory status and ventilator parameters during ACV
-
Support secretion management and airway clearance
Nursing
-
Monitor patient comfort, vital signs, and tolerance during ACV sessions
-
Reinforce ACV use per SLP recommendations
-
Document ACV sessions, patient response, and any adverse events
Medical Team (Physicians and Advanced Practice Providers)
-
Confirm medical appropriateness for ACV
-
Address airway or laryngeal concerns in collaboration with otolaryngology as needed
-
Support escalation or discontinuation decisions based on patient status
Equipment
-
ACV-capable cuffed tracheostomy tube with speech lumen
-
Wall-mounted or portable oxygen or medical air flowmeter
-
Connecting tubing compatible with occlusion valve (thumb port)
-
Continuous monitoring equipment (pulse oximetry)
Suction source capable of providing regulated negative pressure (wall-mounted or portable suction device)
Recommendations
-
Recommendation 1: Patient selection. ACV should be considered only for patients who meet all inclusion criteria and have no contraindications, following interprofessional assessment.
-
Recommendation 2: Interprofessional, SLP-led initiation. Initial ACV trials should be guided by speech-language pathology in collaboration with respiratory therapy and nursing, with medical team oversight.
-
Recommendation 3: Gas flow titration and time limits: Gas flow should begin at low flow (approximately 1 L/min) and be titrated to the lowest effective flow that achieves phonation, with strict time limits and continuous observation.
-
Recommendation 4: Discontinuation and safety safeguards. ACV must be discontinued immediately if the patient demonstrates discomfort, respiratory compromise, subcutaneous emphysema, or signs of air trapping (e.g., inability of delivered gas flow to adequately exit through the upper airway, resulting in pressure build-up). Explicit safeguards must be in place to prevent equipment misconnection, such as labeling the appropriate communication/suction line.
Because the available evidence base for ACV remains limited and heterogeneous, these recommendations should be interpreted as evidence-informed and consensus-supported practice guidance rather than strength-graded clinical recommendations. Clinicians should apply the guidance within the context of individual patient factors, institutional policies, and available resources.
Procedures
Initial ACV Trial
-
Confirm patient eligibility: Confirm that the patient meets all inclusion criteria and has no contraindications to ACV.
-
Verify cuff pressure and positioning: Ensure tracheostomy cuff pressure is within a clinically appropriate range sufficient to maintain seal while minimizing tracheal mucosal injury (20–25 cm H₂O) and remains inflated during ACV. Position the patient upright with the head of the bed elevated whenever possible.
-
Clear the subglottic space: Suction the speech lumen prior to flow delivery to clear pooled secretions above the cuff and reduce coughing or discomfort during ACV initiation.
-
Connect gas flow source: Connect humidified medical air or oxygen to the speech lumen using compatible tubing.
-
Initiate and titrate gas flow: Begin gas flow at 1 liter per minute and gradually titrate to the lowest effective flow that achieves phonation, based on patient tolerance and voice quality. Gas flow should generally not exceed 5 liters per minute unless specifically recommended by speech-language pathology. Higher flow rates may increase the risk of mucosal drying, injury, or air trapping.
-
Monitor patient response: Observe continuously for audible phonation, patient comfort, respiratory stability, and any signs of distress, air trapping, or intolerance.
-
Limit trial duration: Limit the initial ACV trial to a short duration (5–10 minutes).
Ongoing Use
-
ACV sessions should generally be limited to 10–15 minutes per hour as tolerated (to minimize mucosal drying, fatigue, and risk of airway injury). Continuous observation is required during ACV use in acute care settings.
-
Gas flow should be discontinued immediately if the patient experiences discomfort, respiratory compromise, subcutaneous emphysema or signs of air trapping.
Monitoring and Safety Considerations
During ACV, monitor for:
-
Oxygen saturation and respiratory rate
-
Patient-reported discomfort or distress
-
Excessive oral or airway secretions
-
Changes in voice quality suggesting fatigue or laryngeal spasm
Potential complications include discomfort, excessive secretions, stomal air leak, subcutaneous emphysema, laryngospasm, mucosal drying, and air trapping below the vocal folds. Any adverse event requires immediate discontinuation of ACV and notification of the medical team.
Implementation of ACV in resource-limited or non-tertiary care settings may require adaptation based on available equipment, staffing, and clinician training. At a minimum, safe implementation requires a tracheostomy tube with a speech lumen, access to a regulated low-flow gas source, and clinicians trained in tracheostomy care and airway monitoring. In settings where speech-language pathology services are not immediately available, initiation of ACV should occur only under supervision of clinicians experienced in tracheostomy management and airway safety.
Safety Considerations and “What-If” Scenarios
Risk of Misconnection to Pilot Balloon
Accidental delivery of medical air or oxygen into the pilot balloon inflation port instead of the speech lumen represents a high-risk error that may result in acute cuff overinflation, tracheal mucosal injury, airway obstruction, or cuff rupture.
To Mitigate This Risk:
-
The pilot balloon inflation port and the ACV speech lumen must be clearly and permanently labeled at the bedside.
-
Only clinicians trained and deemed competent in ACV may initiate or modify gas flow connections.
-
Prior to each ACV session, a structured verification process must be performed:
-
Visually identify the pilot balloon inflation port and verbally confirm with the team.
-
Measure and document tracheal cuff pressure before and after each ACV session.
-
Visually trace tubing from the gas flow source to the speech lumen connection.
-
Verbally confirm “speech lumen connected” before initiating gas flow.
-
If Misconnection Is Suspected:
-
Immediately discontinue gas flow.
-
Ensure adequate patient ventilation and oxygenation.
-
Assess tracheal cuff pressure and airway patency.
-
Notify respiratory therapy and the medical team.
-
Document the event and initiate institutional safety and incident reporting.
Speech Lumen Occlusion
The speech lumen may become occluded by subglottic secretions, preventing effective phonation.
-
Suction the subglottic port prior to ACV initiation.
-
Flush the subglottic port with saline if needed.
-
If resistance is encountered, ACV should not be forced.
-
Persistent occlusion may require tracheostomy tube exchange per clinical judgment.
-
Thick or tenacious secretions may limit effective ACV use due to repeated occlusion of the speech lumen. Consider reassessment of ACV candidacy if thick secretions persist despite appropriate airway management.
Upper Airway Obstruction or Poor Tolerance
If the patient develops distress, stridor, or absence of phonation despite adequate gas flow:
-
Immediately stop ACV.
-
Evaluate for upper airway obstruction.
-
Consider otolaryngology consultation for airway assessment.
Vocal Fold Injury and Mucosal Drying
-
Use the lowest effective gas flow to achieve phonation.
-
Limit session duration per protocol.
-
Consider humidified gas delivery if prolonged ACV use is anticipated.
Subcutaneous Emphysema or Stomal Air Leak
-
ACV should not be initiated in patients with a fresh tracheostomy (<72 hours since placement) or an unstable stoma.
-
An episode of subcutaneous emphysema does not necessarily preclude subsequent ACV trials once the stoma has further matured and clinical stability has been re-established.
-
New-onset neck swelling or crepitus requires immediate cessation of ACV and urgent medical evaluation.
Abdominal Distention or Aerophagia
-
Gas flow must be turned off when the patient is not actively attempting to speak.
-
Continuous unattended gas flow is prohibited.
Escalation Pathway
Any uncertainty regarding safety, equipment configuration, or patient tolerance should prompt immediate cessation of ACV and escalation to the speech-language pathologist, respiratory therapist, and medical team.
Discontinuation Criteria
ACV should be discontinued if:
-
The patient no longer tolerates the intervention
-
There is a change in airway stability or tracheostomy position
-
The patient progresses to cuff deflation and is able to use more traditional voicing methods
-
A serious adverse event occurs
Documentation
Documentation must be completed in the electronic medical record for each ACV episode and must include, as applicable to scope of practice:
-
Confirmation of patient eligibility and absence of contraindications
-
Tracheostomy tube type and confirmation of an ACV-capable speech lumen
-
Gas flow source (medical air or oxygen), flow rate, and total duration of the ACV session
-
Patient tolerance, voice quality, and communication effectiveness
-
Recommended and optimal gas flow parameters for voicing
-
Names and roles of clinicians or trained individuals present during ACV initiation
-
Any complications, near misses, safety concerns, or deviations from protocol
Any variance from this guidance, including equipment misconnection, premature initiation, or adverse events, must be documented in the medical record and reported in accordance with institutional safety and incident reporting procedures.
Education and Competency
All clinicians involved in ACV must complete institution-approved training and demonstrate competency prior to independent implementation. Ongoing education and periodic review of this guidance are recommended.
External Review and Quality Assurance
This guidance underwent interprofessional review by clinicians with expertise in tracheostomy care, speech-language pathology, respiratory therapy, nursing leadership, and patient safety. Our review focused on clinical feasibility, alignment with current evidence, and mitigation of known safety risks associated with ACV.
Quality Monitoring and Updating
Quality indicators may include time to first speech, patient-reported communication outcomes, ACV-related adverse events, and reported near misses. Service line leaders for nursing, respiratory therapy, and speech-language pathology are accountable for ensuring staff competency, adherence to this guidance, and participation in quality improvement activities.
Limitations and Future Research Needs
The current evidence base for ACV consists largely of observational studies, implementation reports, and narrative reviews, with limited randomized trial data. Future research should focus on prospective comparative studies, standardized communication and quality-of-life outcome measures, and human factors analyses related to equipment design and misconnection risk. The narrative review approach may introduce selection bias; however, the intent of this guidance is pragmatic translation of existing literature and clinical expertise into implementable safety-focused recommendations.
Ethical and Reporting Statement
This manuscript presents evidence-informed clinical practice guidance and does not involve human subjects research as defined by institutional or federal regulations. As such, Institutional Review Board (IRB) review or approval was not required. No identifiable patient data or case reports are included. Reporting of this guidance adheres to the principles of the RIGHT statement.
Conflicts of Interest
All authors declare that they have no conflicts of interest relevant to this work, or they have disclosed all relevant financial and non-financial relationships in accordance with journal and ICMJE requirements.
Funding
This work was supported by the Center for Immersive Learning and Digital Innovation, a Patient Safety Learning Lab advancing patient safety through design, systems engineering, and health services research. This project was funded by the Agency for Healthcare Research and Quality (AHRQ) under grant number R18HS029124. The funding agency had no role in the development of the guidance, interpretation of the evidence, writing of the manuscript, or the decision to submit for publication.
Authorship and Contributions
All authors made substantial contributions to the conception and design of the guidance; synthesis and interpretation of the evidence; drafting and critical revision of the manuscript for important intellectual content; and approved the final version for submission.
Corresponding Author
Vinciya Pandian
The Pennsylvania State University
Ross and Carol Nese College of Nursing
201 Nursing Sciences Building
University Park, PA, USA
Email: vpandian@psu.edu
Phone: +1-443-655-3482